Last Updated May 5, 2026
This article breaks down the science behind what’s happening at the surface level. It explains why traditional approaches fall short and what actually drives lasting results.
If you’ve detailed more than a handful of cars, you’ve seen the same pattern play out. Tires come out deep and dark. Trim looks revived. The vehicle feels finished. Then a week or two later, after rain or a couple washes, the tires start to brown again and the trim fades back toward gray. It shows up on daily drivers, garage-kept cars, even vehicles that were prepped carefully.
At some point, most people assume they did something wrong. In reality, the outcome was built into the product from the beginning.
The Problem Was Never Your Process
In the shop, you can do everything right. You scrub the tire thoroughly, you remove visible browning, you apply evenly, and you let it dry. It still doesn’t last. That’s because traditional dressings and trim restorers are not designed to change the material itself. They are designed to temporarily improve how the surface looks.
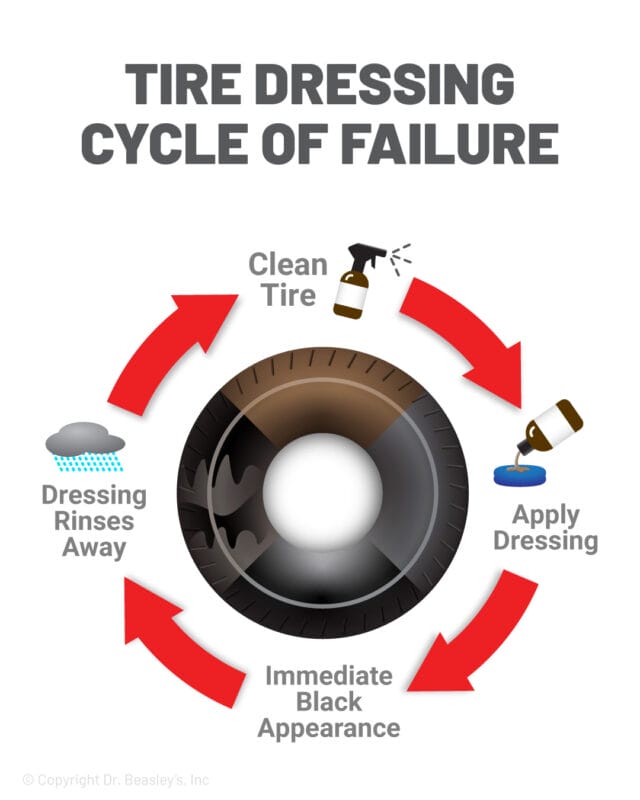
Most of these products rely on silicone oils, emulsions, or solvent carriers that sit on top of the surface. They darken rubber and plastic because they change how light reflects and how the surface wets. But they are not anchored in a meaningful way. They rely on physical adhesion and weak intermolecular forces, not chemical bonding. Once exposed to water, detergents, heat, and road friction, that layer is removed. What’s left is the same surface you started with.
What’s Actually Happening on Tires and Trim
Rubber degradation is not a vague idea. It is a well-understood chemical process driven by oxidation, ozone attack, and mechanical stress. Over time, the polymer chains that give rubber its flexibility begin to break down through chain scission (the degradation of a polymer), reducing elasticity and changing the surface structure. This process accelerates under heat and UV exposure and is a primary driver of aging in elastomers (Progress in Polymer Science, Elsevier).
Tires are also formulated with protective additives, particularly antiozonants (compounds that prevent ozone damage), such as 6PPD, which are designed to migrate to the surface and react with ozone before it can attack the rubber itself. This is a sacrificial protection mechanism. As these compounds move and react, they can contribute to the browning or hazing you see on the surface (Rubber Division, American Chemical Society).
So when you clean a tire and see brown foam, you are not just removing dirt. You are pulling out oxidized material, migrated additives, and embedded contamination. The surface may look clean visually, but chemically it is still an active, changing material.
Exterior plastics behave differently, but the pattern is similar. UV exposure and oxidation degrade stabilizers within the material, leading to fading and loss of color. Again, the issue is not just appearance. It is material degradation at the surface level (Science and Technology of Rubber, Springer).
Why Traditional Dressings Keep Failing
From a formulation standpoint, most dressings are optimized for immediate visual impact. They spread easily, level well, and create gloss or darkening quickly. That is what the user sees.
What they do not do is form a durable structure with the surface.
There is no cross-linked network. There is no meaningful chemical interaction with the substrate. The product sits on top, held in place by surface tension and weak attraction forces. As a result, environmental exposure removes it in a predictable way. Water disrupts it. Detergents emulsify it. Heat accelerates movement and evaporation.
Meanwhile, the underlying material continues to degrade through oxidation and ozone attack. That is why the cycle of apply, fade, reapply keeps repeating. It is not user error. It is the limitation of the mechanism itself (Autoxidation of Polymers, American Chemical Society).

The Shift: From Surface-Level to Surface-Bonding
Changing the outcome requires changing the mechanism.
Instead of placing a temporary layer on top of the surface, a coating system introduces chemistry that can interact at the interface. Many of these systems are built on organofunctional silanes, which undergo hydrolysis and condensation reactions that allow them to attach to surfaces and to each other.
This creates two things at once:
- Attachment to the substrate
- Cross-linking between molecules
The result is a network that is anchored into the microscopic texture of the material rather than simply resting on top of it (Silane Coupling Agents, Elsevier).
| Traditional Trim Restorer | Trim Coating | |
|---|---|---|
| Mechanism | Sits on top | Bonds to surface |
| Structure | Temporary film | Cross-linked layer |
| Effect on plastic | Cosmetic only | Interacts with substrate |
| Adhesion | Weak | Strong |
| UV resistance | Fades quickly | Resists fading |
| Wash resistance | Washes off | Stays in place |
| Appearance over time | Uneven fade | Consistent look |
| Failure mode | Wears away fast | Gradual wear |
| Maintenance | Reapply often | Maintain finish |
| Goal | Quick darkening | Lasting result |
How Cross-Linking Changes Performance
Cross-linking is what turns a temporary film into a durable structure.
In a dressing, molecules are independent. They can move, separate, and be removed easily. In a cross-linked system, molecules are chemically connected, forming a three-dimensional network. This network distributes stress and resists displacement because removing it requires breaking multiple connections, not just wiping away a surface layer (Organic Coatings: Science and Technology, Elsevier).
You can see this difference in real-world use. A dressed tire often shows uneven fading after the first rain, with streaks where water has disrupted the film. A bonded coating maintains a more consistent appearance because the structure remains intact under normal environmental exposure.
Water interaction changes as well. Instead of inconsistent wetting, you get a more stable surface energy, which leads to predictable beading or sheeting behavior.
Why Preparation Determines Everything
Bonding only works if the coating is interacting with the actual substrate.
If there are residual dressings, oils, or degraded material on the surface, the coating will attach to those instead of the rubber or plastic. This creates what is known in adhesion science as a weak boundary layer, and failure will occur at that interface regardless of how strong the coating itself is (Principles of Adhesion and Adhesives, Elsevier).
In practical terms, this is why a surface that “looks clean” is not necessarily ready. True preparation means removing anything that interferes with adhesion so the coating can form a stable, bonded structure.
Why Tires and Trim Require Different Approaches
Tires and trim are not interchangeable surfaces.
Tires are flexible, chemically active, and subject to continuous deformation. Plastics have different surface energies, different degradation pathways, and different requirements for adhesion. A single product trying to handle both often compromises performance.
A system approach allows each component to be designed for its specific substrate. A cleaner that removes residues and resets the surface. A tire coating that can handle flex and exposure. A trim coating optimized for plastic and UV stability. Each step reinforces the others.
What This Looks Like in the Real World
When you move from dressings to a bonded system, the difference shows up after the first wash, not just after application.
You don’t see sling onto paint after driving. You don’t see rapid fading after exposure to rain. The finish holds because it is not dependent on a temporary layer that is constantly being removed.
More importantly, the process changes. Instead of repeatedly restoring appearance, you are maintaining a surface that has already been stabilized.

A Different Category, Not an Incremental Upgrade
This is not a better dressing. It is a different category entirely.
It is the difference between temporarily improving how a surface looks and changing how that surface behaves over time. That distinction is what determines whether results last days or months.
Once you understand the underlying chemistry, the limitations of traditional approaches become obvious. They were never designed to solve the material-level problem.
What Comes Next
We built a complete Tire & Trim Coating System around this approach. It starts with proper surface preparation, followed by coatings designed specifically for rubber and plastic. The goal is not to temporarily darken or shine the surface, but to change how it responds to the environment.
We’ll be sharing application methods, durability testing, and side-by-side comparisons as we get closer to release. If you’ve experienced the cycle of applying and reapplying dressings, this is exactly the problem this system was designed to solve.
Sources
- “The Chemistry of Tire Aging,” Progress in Polymer Science, Elsevier
- “Autoxidation of Polymers and Its Industrial Consequences,” American Chemical Society
- “Antidegradants in Rubber: Mechanisms and Applications,” Rubber Division, ACS
- Science and Technology of Rubber, Springer
- “Ozone Degradation of Elastomers,” Royal Society of Chemistry
- ASTM D1149 – Rubber Deterioration by Ozone Cracking, ASTM International
- Silane Coupling Agents, Elsevier
- “Surface Modification with Organosilanes,” American Chemical Society
- Organic Coatings: Science and Technology, Elsevier
- Principles of Adhesion and Adhesives, Elsevier





Questions? Comments?
Email Us